ICSR Management
Streamline your pharmacovigilance workflows with our expert
individual case safety report (ICSR) services, ensuring rapid,
accurate, and compliant case safety processing for medicinal products in line with global regulatory requirements.
individual case safety report (ICSR) services, ensuring rapid,
accurate, and compliant case safety processing for medicinal products in line with global regulatory requirements.

Why Structured ICSR Workflows Are Critical for Compliance
Managing product safety data is a race against time and complexity. When a team manually processes data from diverse sources, they often miss deadlines. Structured workflows are critical to address key challenges, such as data fragmentation and changing regulatory standards. They also ensure that reporting requirements are met and that audits are always ready to provide access to documentation.
Our Individual Case Safety Report Services: Scope and Deliverables
We provide comprehensive support for every individual case safety report, ensuring precision from initial detection to final submission. Our team optimizes your data management to guarantee global compliance and enhanced patient safety.
End-to-End Case Intake and Triage
We manage the full lifecycle of an individual case safety report for pharmacovigilance, from multi-channel data collection to finalisation. Our experts perform rapid triage, prioritization and identification of duplicates to ensure your safety database remains clean, organized, and audit-ready.
AI-Assisted Case Processing and Medical Coding
Leveraging the DrugCard technology platform, we accelerate data entry through automated extraction. Our specialists perform precise MedDRA coding and the WHO Drug Dictionary application to maintain the highest standards of accuracy. We process technical files in accordance with implementation guides to ensure compliance with standards for every case.
Quality Review and Case Validation
Quality is the most important part of how we do business. Every case undergoes a thorough secondary review by experienced PV professionals to ensure that all aspects are correct. This eliminates mistakes before the report reaches its final destination.
Timely Submissions to Regulatory Authorities
We ensure your safety data meets strict E2B(R3) standards for seamless electronic submission. Our team monitors global regulatory requirements and timelines to guarantee that all expedited filings are sent to health authorities without delay.
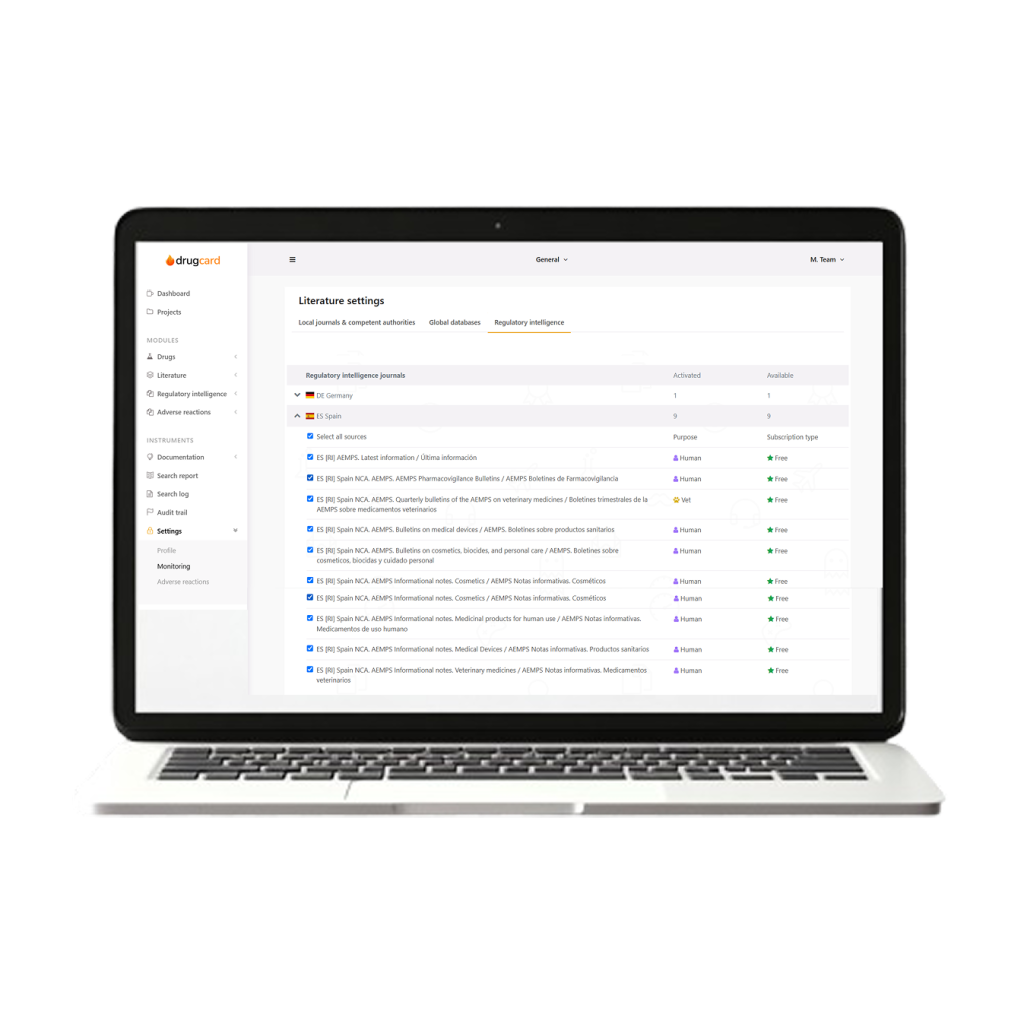
Literature Monitoring and Follow-Up Management
Our team proactively identifies potential signals through systematic literature monitoring and manages the follow-up process for all identified information. This ensures that each case is as complete as possible for effective safety management and regulatory actions.
DrugCard Platform in Numbers
121+
Countries
Medical journals coverage
Medical journals coverage
2200+
Local journals
Continuously monitored
Continuously monitored
~2
Weeks
New country Easily Added
New country Easily Added
How Our SaaS Platform Enhances ICSR Workflows
By combining the precision of the DrugCard SaaS platform with our expert medical team, we deliver an unmatched hybrid solution to provide access to real-time data. The platform pulls data from local, global literature and regulatory announcements, while our experts provide the human oversight needed for narrative writing in complicated medical evaluations. This synergy improves efficiency, reduces mistakes, and ensures your process stays in line with global compliance standards.

Benefits of Outsourcing ICSR Management
- Reduce overhead by eliminating the need for extensive in-house pharmacovigilance infrastructure.
- Effortlessly manage fluctuating case volumes without delays in recruitment.
- Access deep expertise in E2B(R3) standards and global reporting timelines.
- Free your internal team to prioritize benefit-risk evaluation and strategic regulatory actions across your entire product portfolio.”
Who Can Benefit From Our ICSR Solutions
Our ICSR management solutions are made for pharmaceutical companies and clinical research organizations (CROs) that want to make their safety data processing easier. Our platform and expert team can help you keep up with local and global literature monitoring or run complicated clinical trials. They can also make sure that you stay in compliance and keep patients safe in all markets.

Supported Case Types
Our ICSR management services cover a wide range of safety data sources and adverse event reporting channels. We expertly handle spontaneous reports, clinical trial events, and individual case safety events identified through literature. Whether dealing with drugs or vaccines, our team ensures every ICSR report undergoes a rigorous medical review and is meticulously processed for regulatory submission.
ICSR Management Workflow: How We Deliver Results
Our streamlined process integrates the DrugCard platform with expert medical oversight. Data from multiple sources is queried and submitted using automated processes that include AI-supported coding and thorough human validation. The end product is a case that meets the standards for high-quality reporting and appropriate regulatory submission, delivered to the appropriate international regulatory agency database as quickly as possible.
Compliance and Data Security Standards
Patient privacy is extremely important to us. All of our services follow E2B (R3) electronic reporting guidelines and GVP module requirements. We will routinely encrypt the transfer of personal information as well as provide strong access controls to keep your information safe. In addition, all of our services comply with international data protection laws.

Why Choose Us for ICSR Management
When you choose DrugCard, you work with a team that knows how to use technology to keep an eye on drugs. We also offer a full-service solution that can grow with your needs and cut down on manual work by up to 80%. With our extensive knowledge of regulations and cutting-edge AI extraction, you can be sure that your safety reporting is compliant, and you can process everything faster and at a lower cost.
Have a question? Just fill out the form and we will contact you within 60 min
- Book a meeting for the demonstraion
- Anwers your additional questions
- Implenetation DrugCard to your pharmacovigilance system
- Tariff plans, pricing, addional features
