AI-Powered Literature Monitoring
Key Benefits of Our AI-Driven Literature Monitoring Platform

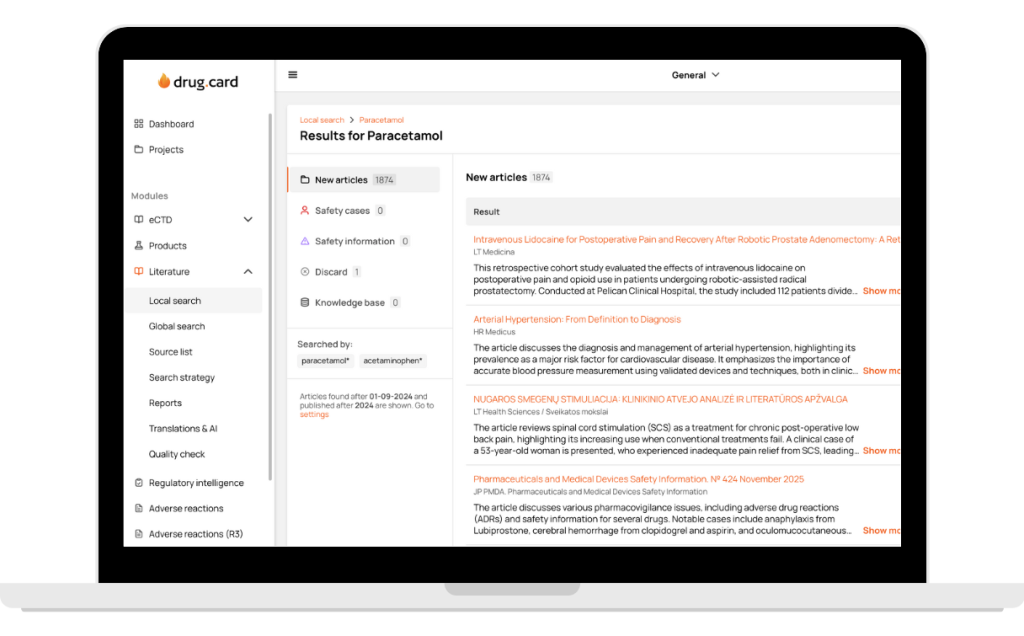
Complete Global & Local Coverage
All medical sources in one platform — from global literature full-text monitoring to 121 countries and 2200+ local sources. Every client gets a tailored source list, country-specific recommendations, and fast addition of any journal without bureaucracy.

Higher Impact With the Same Team
Automation removes repetitive literature screening so your experts can focus on real PV work — signal detection, risk assessment, drug safety documentation, and scientific evaluation.

Zero Missed Publications, Zero Compliance Risk

Full Visibility and Built-In Documentation

Data Integrity and Security

Always-On Access, Anywhere
DrugCard Platform in Numbers
Medical journals coverage
Continuously monitored
New country Easily Added
Comprehensive Feature Set for Literature Screening
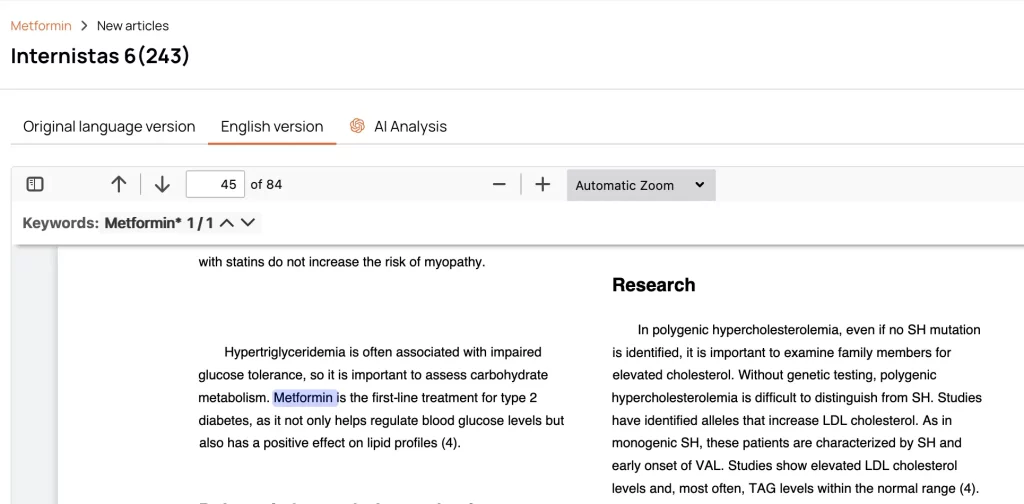
AI Article Summaries
AI Pre-Assessment of Safety Relevance
Artificial intelligence identifies whether an article contains any safety-relevant references, safety signals, and prioritizes it.
One-Click Case Creation From Literature
Convert an article into an individual case safety report in a single click. Fully integrated with the DrugCard adverse event database.
Automatic Translation of Local Sources
Validated, Audit-Ready Platform
Flexible Project Configuration

A Shift From Manual Reading to Automated Medical Literature
The problem with manual screening
Manual literature review consumes hours of repetitive literature surveillance and regulatory reporting, increases the risk of human error and burnout, and makes traceability difficult.
How DrugCard solves it
How It Works
Our Case Studies
Why Leading Pharmacovigilance Teams Choose Us
Unmatched Expertise in Literature Screening
Full Compliance with GVP & Data Security